Dear Customers,
A few shipments arrived this week. If you log into your account at www.toywonders.com, before clicking on any of the links below, approved wholesale accounts will see wholesale pricing.
Forgot your password?
Having trouble viewing this page
Missed a previous week's newsletter?
DIECAST Collectible Model Cars And More
|
Image |
Item# |
Description |
Stock Status |
|
5355DF |
|
New |
|
5361DF |
|
New |
|
5365DPR/P |
|
New |
|
5365DPR/R |
|
New |
|
22076WBK |
|
New |
|
22076WSV |
|
New |
|
22083WBK |
|
New |
|
22083WBN |
|
New |
|
22086/4D |
|
New |
|
22086W/BU |
|
New |
|
22086WR |
|
New |
|
22089W/OR |
|
New |
|
22089WSV |
|
New |
|
22414CWR |
|
New |
|
22414CWW |
|
New |
|
22414H/WR |
|
New |
|
22414HWW |
|
New |
|
22433/4D |
|
New |
|
22433WW |
|
New |
|
22433WYL |
|
New |
|
22434/4D |
|
New |
|
22434WBK |
|
New |
|
22434WR |
|
New |
|
22448WBU |
|
New |
|
22448WR |
|
New |
|
22451WR |
|
New |
|
22501/4D |
|
New |
|
22501W/OR |
|
New |
|
22501W/W |
|
New |
|
22502W/BU |
|
New |
|
22502W/SV |
|
New |
|
24002/4D |
|
New |
|
24031/4D |
|
New |
|
24031WGY |
|
New |
|
24042WR |
|
New |
|
24042WYL |
|
New |
|
24045/4D |
|
New |
|
24045WBK |
|
New |
|
24045WW |
|
New |
|
24046/4D |
|
New |
|
24046WBN |
|
New |
|
24046WBU |
|
New |
|
29393WBK |
|
New |
|
29393WR |
|
New |
|
29397WCM |
|
New |
|
29397WR |
|
New |
|
42391D |
|
New |
|
43624D |
|
New |
|
43641D |
|
New |
|
43667D |
|
New |
Please do not reply to this email address.
Any questions or comments, please email us at [email protected].
To unsubscribe to this newsletter, send an email to [email protected] and put the word 'unsubscribe' on the subject line.
Thank
you
Lu
Toy Wonders, Inc.
www.toywonders.com
201-229-1700

God and the Art of Toy and Diecast
Marketing
Something is Awry with the Timeline (part 5)
By L. S. Su and Robert Doolan
It's been a long and cold Winter
in the Northeast. Experience negative to single digit temperatures
for a few weeks and then suddenly 30 degree seems like a heat
wave. Seriously, I had multiple people telling me at 32 degrees
it was "warm outside." I've noticed that my southern
Californian friends and relatives have a very different idea
what cold is. Freezing to them is when the temperature drops
to below 75 degrees. But hot and cold is all relative right?
So here is an interesting question:
Does TIME work the same way? Is time relative to the beholder?
Does time move faster for some, but slower for others?
We might perceive the passing of
time differently, but in reality you and I have exactly the
same amount of time to do stuff each day. Timelines are not
relative (i.e. this timeline is true for you, but false for
me); However, most of academia's timelines are relative to the
tools they use. Let me explain.
When it comes to dating very old
objects, academia appears to only have one tool in their tool
box; it's called radiometric dating. In the last article I talked
about the most popular type of radiometric dating called carbon
14 (C14 dating). But I pointed in
my last article, out that one of the biggest problems with
any type of radiometric dating is great number of assumptions
that need to be made. One very big assumption is that the starting
levels of that radioactive material in the object you are measuring
is known. Another assumption is that the departure rate of the
radioactive isotope (rate of decay) is steady. Exactly after
that radioactive element's half life, only half of that isotope
will remain in that object -at least that is how the theory
goes. So the breakdown of radioactivity is a linear progression?
This is a valid assumption to make? Even if we know the starting
levels (which at best is an estimate) and even the rate is a
constant linear progression, I see a fundamental problem with
the half life theory.
Say we know the starting quantity
of something like a bowl of Cheerios made from genetically modified
wheat (so maybe there really is some radioactivity going on).
Pretend that bowl of Cheerios represents an animal fossil. When
it dies it stops taking in C14 (Cheerios). So the way half-life
theory works is that every time we take half the quantity of
Cheerios out of the bowl, how long ago that animal died increases
(i.e. its half life). So in our life time (in a few minutes)
we can continue to take half the quantity out of the bowl and
eventually get to either one (1) or one and a half (1.5) Cheerios
left. But when we get to this point, most scientists don't conclude,
"Okay we've hit the limit, and now can calculate how long
ago this animal lived."
The problem with half-life theory
is that you can continue to divide a quantity for a very long
time right? You go from one (1) Cheerio to half, to a quarter,
to an eighth, and so on. In theory, you can do this forever.
Granted that this quantity won't do a good job in filling our
stomachs, but 10 trillion years from now, we can still be dividing
that original quantity of Cheerios. Since in theory you can
continue to take half off something forever, then half life
theory tells me that objects can be infinitely old. But that
defies common sense.
Nobody in academia wants to write
a article proposing that this bowl of Cheerios (fossil remains)
is older than the Big Bang. So what I feel academia does at
that point is to construct some artificial ceiling to the half
life theory (e.g. carbon dating is only good to the 9th or 10th
recursion). And then state something like, "Its because
we don't have the tools to measure such a miniscule quantity."
But then what I find really interesting is at this point, instead
of saying "we've now hit the limit of how long ago this
animal walked the Earth.", they switch to another radiometric
dating method (one with a longer half life) and start measuring
inorganic matter around that fossil.
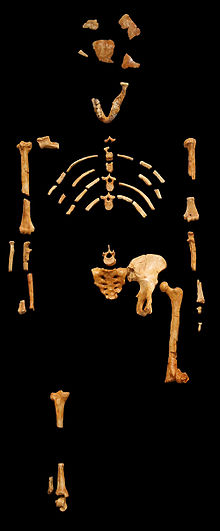
So for example, probably the most
well know find were bones found at the bottom of a cliff in
Africa and they were given the name Lucy. Radiometric dating
puts these bones at approximately 3.2 million years old. But
radiometric dating wasn't applied to the bones, but to the soil
around the bones. What Scientist try to look for is volcanic
ash in the soil, because they think they know the starting amounts
of radioactive argon in ash/lava. First of all, I question if
this is a valid assumption to make that the age of the fossil
is the same age as the soil layer the fossil was found in. Say
an earthquake occurred and you and I fell down into a chasm.
Measuring the age of the soil near our bones is a good way to
figure out when you and I ate Cheerios? Isn't it reasonalbe
that the soil needs to be much much older than the fossil? Moreover,
water has proven to invalidate radiometric dating results. Also
water can displace soil and bones (and even houses via Hurricane
Sandy).
But what specifically in soil are
scientist looking for? The answer is volcanic ash, because they
think they know the starting levels of the radioactive isotope
potassium-argon. This is not true. Multiple scientists have
proven the potassium-argon levels in lava can be different,
even in the same eruption.

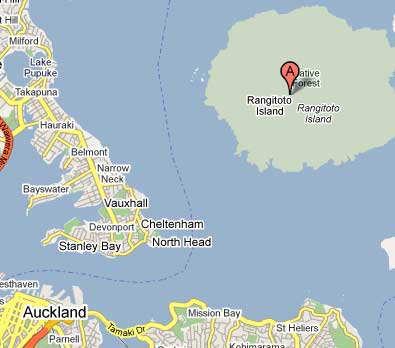
Among impressive volcanic scenery
in northern New Zealand lies the city of Auckland. The district
is known for its volcanic cones. In fact, there are more than
50 recognized small volcanoes in the city and surrounding areas.
But the largest volcano by far in Auckland is also the youngest.
It is called Rangitoto. How young is this youngest volcano?
Now your problem starts. Rangitoto is generally regarded as
young for several reasons. Evidence based on botany and geomorphology,
and a hint from Maori legend that the name can mean ‘red
sky’, contribute to a common acceptance that Rangitoto
is youthful. Some of the lavas (scoria) have no vegetation,
and seem to be no more than a few hundred years old.
Conflicting dates
In the late 1960s, scientists from
the Australian National University in Canberra dated numerous
volcanoes in Auckland using the potassium-argon method. Ten
samples from both vegetated and unvegetated lava on Rangitoto
were dated. Results seemed to show that Rangitoto was not a
few hundred years old as it appeared to be. Ages from the 10
samples ranged from 146,000 years up to almost half a million
years! So how old is Rangitoto? A couple of hundred years? Or
half a million? The scientists took a sample of wood from beneath
some Rangitoto lava and dated it by the carbon- 14 method. The
wood gave an age of only 225 years (plus or minus 110 years)—which
potentially puts it in the lifetime of George Washington and
German composer Johann Sebastian Bach. This is about the age
all evidence points to except potassium-argon dating. If lava
which is little more than 200 years old can be wrongly dated
at up to 465,000 years by the potassium-argon method, could
potassium-argon dating always be wrong?
Wrong every time
The scientists who did the Rangitoto tests dated
16 volcanoes in all. Eleven of these were able to be compared
with carbon-14 dates. In every case the potassium-argon dates
were clearly wrong to a huge extent. Similar conflict was found
by researchers in Hawaii. A lava flow which is known to have
taken place in 1800-1801—less than 220 years ago—was
dated by potassium-argon as being 2,960 million years old. If
the real dates were not fairly well established by other means,
who could have proved that the potassium-argon dates were so
wrong? So how do you date a volcano? The lesson seems to be
that how ever you date it, don’t count on the potassium-argon
method.
Please note that these results do not stand or
fall on the dating of xenoliths (something that pre-existed
lava and wasn't destroyed by the intense heat), as at least
one critic has wrongly claimed, because in every case in which
the potassium-argon dates could be compared with the carbon-14
dates, the potassium-argon dates were wrong.
References:
Robert Doolan, "How do you date a New Zealand
Volcano?" http://creation.com/how-do-you-date-a-new-zealand-volcano
Ian McDougall, H. A. Polach and J.J. Stipp, ‘Excess
radiogenic argon in young subaerial basalts from the Auckland
volcanic field, New Zealand’, Geochimica et Cosmochimica
Acta, Vol.33. 1969, pp. 1485-1520.
Carbon-14 dating is regarded by creationists as reasonably reliable
for recent objects. For explanation see The Answers Book, pp.43-SO.
(See ad p.40 this issue.)
J. G. Punkhouser and J.J. Naughton, ‘He and Ar in ultramafic
inclusions’, Journal of Geophysical Research, Vol.73,1968,
pp. 4601-4607.
Back to the top