Dear Customers,
A few shipments arrived this week. If you log into your account at www.toywonders.com, before clicking on any of the links below, approved wholesale accounts will see wholesale pricing.
Forgot your password?
Missed a previous week's newsletter?
DIECAST Collectible Model Cars And More
|
Image |
Item# |
Description |
Stock Status |
|
12855 |
Greenlight Starsky & Hutch - Ford Grand Torino Hard Top (1974, 1:18, Red w/ White Stripe) 12855 |
New |
|
18213 |
Greenlight Bewitched - Chevrolet Camaro Convertible (1969, 1:24, Green) 18213 |
New |
|
18211 |
Greenlight Men In Black 3 - Ford Taurus Sho Hard Top (2012, 1:24, Black) 18211 |
Restock |
|
21956P |
Tomy Johnny Lightning - The Dukes of Hazzard Dodge Monoco Rosco Patrol Car (1974, 1:18, White) 21956P |
New |
|
1800809 |
Acme - Fuel Altered Dragster Pure Heaven II (1:18) 1800809 |
New |
|
1805003 |
Acme So-Cal - Ford Coupe Hard Top (1932, 1:18, Red & White) 1805003 |
New |
|
40300/31 |
Castline M2 Machines Auto-Dreams Premium - Dodge Challenger R/T Hemi Hard Top & Plymouth Hemi Cuda Hard Top (1970/ 1971, 1:24, Black) 40300/31 |
New |
|
C142823 |
Lionel Nascar - Chevy Impala Mobil 1 Tony Stewart #14 Race Car (2012, 1:24) C142823 |
New |
|
C142866 |
Lionel Nascar - Chevy Impala Mobil 1 Tony Stewart #14 Race Car (2012, 1:64) C142866 |
New |
|
C242823 |
Lionel Nascar Hendrick - Chevy Impala DuPont Jeff Gordon #24 Race Car (2012, 1:24) C242823 |
New |
|
C242866 |
Lionel Nascar Hendrick - Chevy Impala Pepsi Max Jeff Gordon #24 Race Car (2012, 1:64) C242866 |
New |
|
C882823 |
Lionel Nascar Hendrick - Chevy Impala Diet Mtn Dew Dale Earnhardt Jr. #88 Race Car (2012, 1:24) C882823 |
New |
|
76256 |
Motormax - Iron Choppers Motorcycle (1:18, Asstd.) 76256 |
New |
Lionel Trains & Diorama
Please do not reply to this email address.
Any questions or comments, please email us at [email protected].
To unsubscribe to this newsletter, send an email to [email protected] and put the word 'unsubscribe' on the subject line.
Thank you
Lu Su
Toy Wonders, Inc.
www.toywonders.com
201-229-1700

God and the Art of Toy and Diecast Marketing
A Mystery with Names (radiometric dating)
By L. S. Su
Last time we looked at the issue where science leads us to believe that man is millions of years old. But the Bible, through the use of these mysterious names of individuals, would lead us to believe that man is only thousands of years old.
Right now the biggest tool in the scientists' tool box for figure out how old stuff is in the world is is called radiometric dating. More specifically, radiometric carbon dating (or radiocarbon dating) is used to date organic material. Carbon dating only works for stuff that once was alive. So carbon dating can be used on stuff like bone, teeth, horns, hair, coral, plants, trees, people, and even on that annoying purple dinosaur called Barney. And whatever happened to that cantankerous space alien puppet ALF?
Now I know it's been a very long time since you brushed up on terms like "carbon-14 dating". As you probably know carbon dating is something very different than speed dating, but if you are doing either, you are looking at something getting old. When we we taught how radiocarbon dating works in high school, it was taught that this was a very accurate way to measure the age of dead stuff, but for carbon-14 dating to date things accurately, it needs to make some assumptions. And I know that those assumptions were either glossed over or never presented. Now I know for us marketers of toys and diecast model cars, carbon dating just doesn't come up in conversation much. So here is my three paragraphs on how it works.
Living organisms while they are alive accumulate carbon in their cells. Plants take it in in the form of carbon dioxide and through photosynthesis the carbon gets incorporated into the plants cells. Then in turn humans and animals ingest the plants and the carbon goes into their cells. Another way for humans and animals to get carbon into them is by ingesting animals and birds, who had dined on plants. I guess if you want to be technical and scientific about it, a third way for humans to get more carbon in them is for humans to eat humans. But most human cultures seem to frown on that type of carbon transfer.
As long as the organism is living, it's accumulating carbon. By the way scientists think carbon is the 4th most common element known to man. The two types of carbon atoms that interest scientists the most are carbon-12 and carbon-14. Carbon 12 has 6 protons, 6 neutrons, and 6 electrons; it the most common and considered stable. But carbon-14 has 6 protons, 8 neutrons, and 6 electrons (adding the neutrons and electrons get you to 14) and is considered unstable (radioactive). As time goes on, carbon-14 wants to shed those neutrons in order to become more stable (and be like its brother carbon-12).
Our atmosphere contains proportion both carbon-12 and carbon-14 in a known proportion. So when the organism is alive it accumulates carbon in the same proportion (i.e. most of the time). When that organism dies, no more accumulation occurs and since the carbon fourteen decays at a known exponential rate. This then makes it possible to calculate the approximate age of the organism based on how much Carbon-14 is left. The less carbon-14 isotope they find, the older the object. It should be noted that the half life of carbon-14 is 5730 (give or take 40 years). So that means after 5730 years, the sample will have half of the carbon-14 that it started with. Scientists seem to agree that the current carbon dating methodology is good only to about 55,000 years; and once past that age, the amount of carbon-14 left in the sample is so minute that you would need to use another dating method. So when scientists claim that the Lucy fossil is 3.2 million years old, they are NOT using carbon dating to come up with that age.

But for radiocarbon dating to work properly, it must assume three things:
1) Carbon-14 decays at a fixed known rate.
2) Living organism while it was alive and after it died, did not come into contact with anything in its environment that would affect the amount of carbon-14 it contains.
3) The proportion of carbon-14 to carbon-12 found in the atmosphere today is about the same ratio as it was several thousand years ago.
Assumption #1 Carbon-14 decays at a fixed known rate:
For you to assume that something decays at a constant rate, you need to first assume that time is constant. But there are some scientists like Einstein who claimed this was not true. However for the sake of this article (and my lack of knowledge of the theory of relativity and quantum physic), lets assume the rate of decay for carbon-14 is constant and is a true assumption.
Assumption #2 The once living organism sample did NOT get tainted somehow
Scientist must like assumption #2, because it provides them a way out in explaining why their age dating was incorrect or even when another lab comes up with a very different age date conclusion from theirs. Here are two known environmental occurrences that would affect the age dating with the use of radiocarbon dating.
The geologist F.W. Shotton from the University of Birmingham claimed that radiocarbon dating makes sea plants and certain sea creatures much older than they really are. "The hard-water effect is a recognized source of error in radiocarbon dating. It causes ages to be over-assessed and arises when the material to be dated, such as mollusc shell or plant, synthesizes its skeleton under water and so uses bicarbonate derived in part from old, inert sources. It is usually stated that the maximum possible error is equivalent to the half-life of 14C, 5,570 yr (Libby scale), but it rarely amounts to as much as this. Moreover, in an isolated dating it is not only impossible to evaluate, but even to know whether there is any over-estimation, but if the sample consists of tree wood, or the leaves, twigs or seeds of wholly terrestrial plants, then it can be assumed that no hard-water error is present."
So basically, this scientist is claiming that it depends where the sea plant or sea creature gets its carbon from. If it gets it from old inert sources, carbon age dating will give misleading results. The lack of carbon-14 in the plant or animals cell is going to cause carbon dating to conclude that it's much older than it really is. So sea water can affect the dating process.
Volcanic activity also has a direct effect on carbon radiometric dating. "Plants which grow in the vicinity of active volcanic fumeroles will yield a radiocarbon age which is too old. Bruns et al. (1980) measured the radioactivity of modern plants growing near hot springs heated by volcanic rocks in western Germany and demonstrated a deficiency in radiocarbon of up to 1500 years through comparison with modern atmospheric radiocarbon levels. Similarly, this effect has been noted for plants in the bay of Palaea Kameni near the prehistoric site of Akrotiri, which was buried by the eruption of the Thera volcano over 3500 years ago (see Weninger, 1989). The effect has been suggested as providing dates in error for the eruption of Thera which has been linked to the demise of the Minoan civilisation in the Aegean. One modern plant growing near the emanations had an apparent age of 1390 yr. The volcanic effect has a limited distance however. Bruns et al. (1980) found that at 200 m away from the source, plants yielded an age in agreement with that expected."
When I read the article, I couldn't figure out if the "200 m" meant 200 meters or 200 miles. But organic stuff living within 200 meters from a volcano probably wouldn't live that long or at least be pretty sick. But nevertheless, scientists are discovering more factors that could affect (taint) the sample and thus make the carbon age dating far older than it really was. But for sake of this article, let's also assume that assumption #2 is true and that scientists have figured out a way to determine whether their sample grew up outside the 200 m radius of a volcano and that this creature wasn't in the habit of dining on certain sea plants and creatures. Let's also assume the have figured out a way to accurately calibrate their instruments to take into account all the known occurrences to taint samples.
Assumption #3 The proportion of carbon-14 to carbon-12 found in the atmosphere today is approximately the same ratio it was several thousand years ago.
When scientist measure how much carbon-14 is left in an organic object, they are comparing the carbon-14 to carbon-12 ratio in that sample to the carbon-14 to carbon-12 ratio in the atmosphere. What they didn't teach us in high school is the exact ratio of carbon-14 to carbon-12. Is it 1 part carbon-14 to 2 parts carbon-12? Or is it something smaller like our scale model cars, 1:24 or 1:18? Or perhaps even smaller 1:87?
The NDT Resource Center says, "The ratio of normal carbon (carbon-12) to carbon-14 in the air and in all living things at any given time is nearly constant. Maybe one in a trillion carbon atoms are carbon-14." Note how the writer of the article assumes that the ratio "at any given time" is nearly constant, but doesn't seem sure what the ratio is "Maybe one in a trillion" Maybe?. The Argonne National Laboratory says the ratio is "about two trillionths". About? One trillionth versus two trillionths, such a tiny difference right? No it's a huge difference one number is twice the size of the other! This is your starting ratio that scientists assume the fossil had when it died.
So the point I am making is that scientists are measuring very miniscule amounts of carbon-14 in their samples (and definitely not using a bathroom scale) and the absence of the tiniest amounts of carbon-14 could have a huge affect on their age estimates. So back to the assumption, The proportion of carbon-14 to carbon-12 found in the atmosphere today is approximately the same several thousand of years ago." Is this a reasonable assumption to make? Before you can make such a conclusion, a reasonable question to ask is how does carbon-14 get into the atmosphere in the first place?
According to the Argonne National Labs article, "Carbon-14 is produced naturally in the upper atmosphere by the reaction of neutrons originating from cosmic rays with nitrogen and, to a lesser extent, with oxygen and carbon". Ahh........so cosmic rays interacting with nitrogen in our atmosphere creates this radioactive carbon-14. So for assumption #3 above to be true, scientist must also conclude that the amount of cosmic rays and nitrogen in our atmosphere has also stayed constant since life existed on Earth. To conclude that the amount of cosmic rays hitting our atmosphere has been constant a reasonable assumption?
In Arthur Strahler's book Science and Earth History, he says, "A study of the deviations from the accurate tree-ring dating sequence shows that the Earth's magnetic field has an important effect on carbon-14 production. When the dipole moment is strong, carbon-14 production is suppressed below normal; when it is weak, carbon-14 production is boosted above normal. What the magnetic field does is to partially shield the earth from cosmic rays which produce carbon-14 high in the atmosphere."
So what this scientist is saying is concluding is that the stronger the Earth's magnetic poles are, the less likely Earth's atmosphere is able to produce carbon-14. So if this is true, then another assumption for radio carbon-14 dating to be accurate is that the strength of Earth's magnetic field (caused by the two opposite poles) has stayed constant since life came into existence on Earth. Is this reasonable assumption to make?
The prevailing answer to this question in the scientific community is NO. One article from the Nasa Science News says, "Our planet's magnetic field is in a constant state of change, say researchers who are beginning to understand how it behaves and why." Furthermore, there is more and more evidence that points us to the fact that the Earth's magnetic field is weakening. National Geographic published an article that said, "Earth's magnetic field is fading. Today it is about 10 percent weaker than it was when German mathematician Carl Friedrich Gauss started keeping tabs on it in 1845, scientists say."
If Earth's magnetic field was stronger thousands of years ago, there would have been less carbon-14 in the atmosphere. This would mean plants and animals would have had far less carbon-14 to begin with (back then). This would then mean current carbon-14 dating methods are misdating the age of fossils and making them much older than they really were.
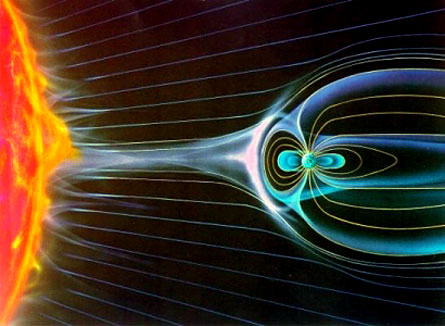
So in regards to assumption #3 (the proportion of carbon-14 to carbon-12 found in the atmosphere today has stayed relatively constant since life forms existing on Earth) to be true, requires the Earth's magnetic field to have stayed relatively constant too. I personally reject this assumption. Do you realize that without Earth's magnetic field, all life forms on Earth would be gone? The stronger the Earth's magnetic field the more it repels harmful cosmic and ultraviolet rays which have proven to damage healthy cells and stuff in our cells (like DNA.). A weaker magnetic field would correspond to increase rates of damaged cells (cancer) and genetic defects.
In short a conclusion that Earth's magnetic field was much stronger in the past than it is today, would put into question one of carbon dating's major assumptions. If the third assumption was false, then this would require scientists to totally revaluate how carbon dating works. But that would be a lot of extra work and everyone is pretty busy now.
However, there is Biblical evidence that something drastically change in Earth's environment after the great flood. Next time let's look at some pre-flood and post-flood passages and see if any of them can be correlated to a weakening of Earth's magnetic field.
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Sources:
Strahler, Arthur N. 1987. Science and Earth History Prometheus Books, 59 John Glenn Drive, Amherst, New York 14228-2197 552 pages (double column)
John Arthur Harrison, Ph.D. Vision Learning Carbon Cycle, What Goes Around Comes Around, http://www.visionlearning.com/library/module_viewer.php?mid=95
Shotton, F.W. Nature :240 An Example of Hard-Water Error in Radiocarbon Dating of Vegetable Matter, 460 - 461 (22 December 1972); doi:10.1038/240460a0
NDT Resource Center, Carbon-14 Dating, http://www.ndt-ed.org/EducationResources/HighSchool/Radiography/carbon14dating.htm
The Argonne National Labartory EVS, Human Health Fact Sheet, August 2005, Carbon-14, http://www.ead.anl.gov/pub/doc/carbon14.pdf
NASA Science News, 2003, Earth's Inconstant Magnetic Field, http://science.nasa.gov/science-news/science-at-nasa/2003/29dec_magneticfield/
National Geographics News, John Roach Sept. 9, 2004, Earth's Magnetic Field Is Fading, http://news.nationalgeographic.com/news/2004/09/0909_040909_earthmagfield.html
--------------------------------
Back to the top